ntP2O5 on treatment with exces s of H2O followed by excess of NH4OH forms (NH4)2HPO4. If hundred gram of (NH4)2HPO4 is formed then find out the mass of p 2 o5 initial
![PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e9e458615e33c40d94b80a3189efa26d438c8a39/4-Figure2-1.png)
PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar

One mole of P2O5 undergoes hydrolysis as P2O5 + H2O⟶H3PO4 The normality of the phosphoric acid formed is (The volume of solution is 1 L)
Complete and balance the following equation (i) P2O5 + H2O → ............ (ii) S + O2 → ................ - Sarthaks eConnect | Largest Online Education Community
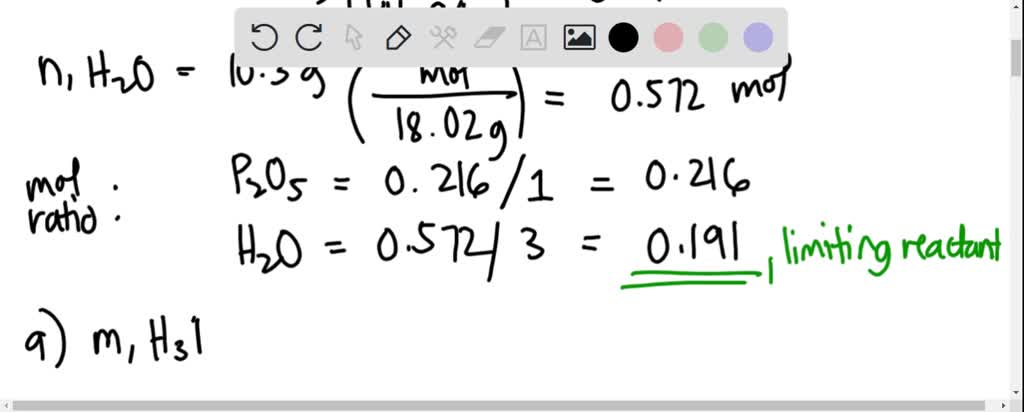
SOLVED: Identify the limiting reactant in the reaction of diphosphorus pentoxide and water to form H3PO4, if 27.4 g of P2O5 and 14.2 g of H2O are combined. Determine the amount (in

SOLVED: The oxidation of phosphine (PH3) to phosphorus pentoxide (P2O5) is given by the chemical reaction.PH3 +O2 —> P2O5 + H2Ob. How many grams of O2 will react completely with 35.0 grams

Solubility study at high phosphorus pentoxide concentration in ternary system CaCO3+P2O5+H2O at 25, 35 and 70 °C - ScienceDirect