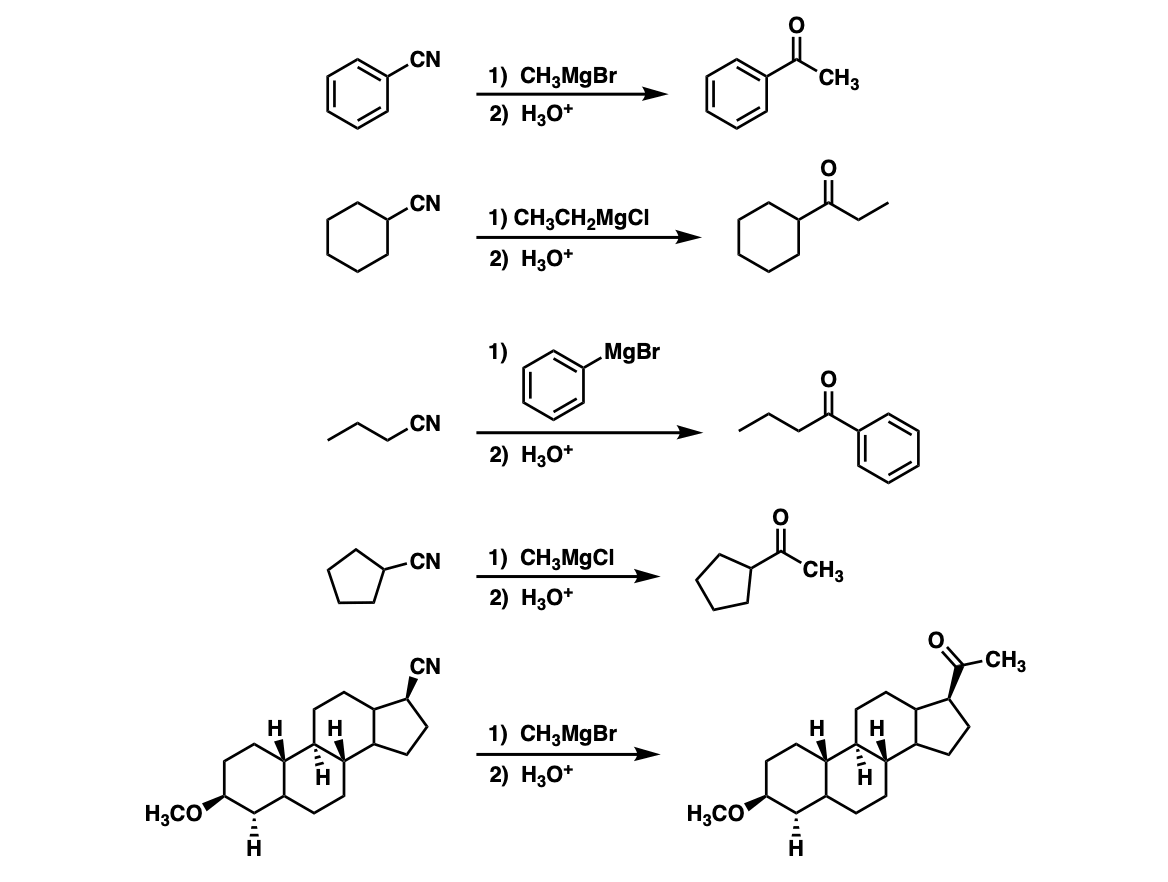
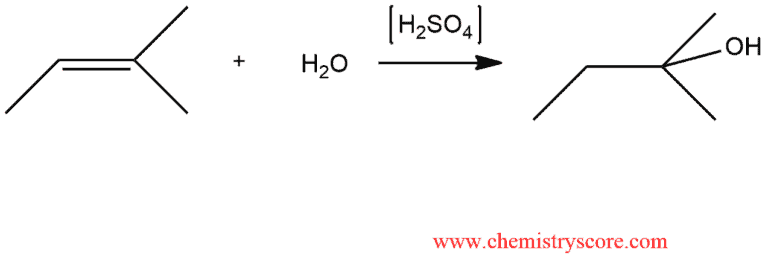
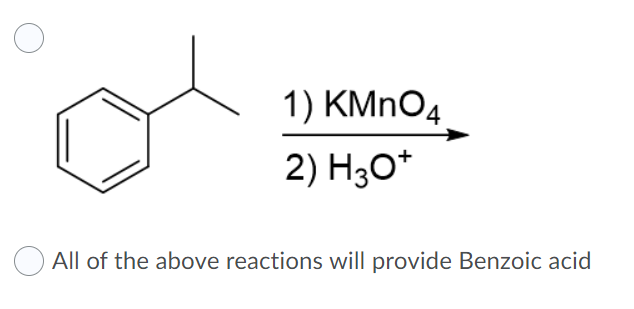
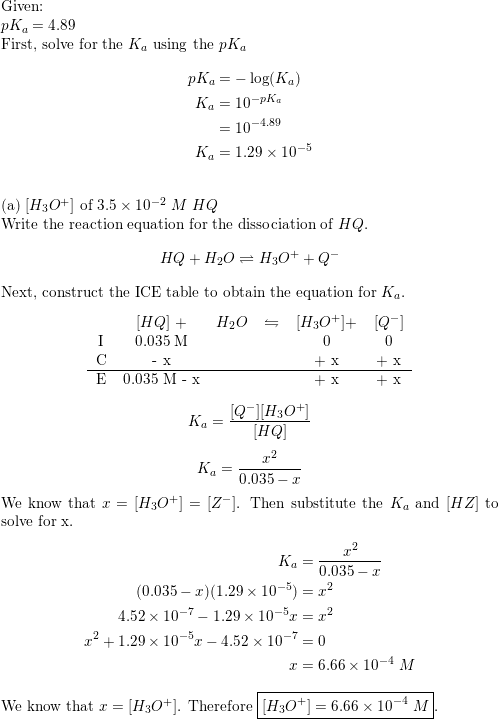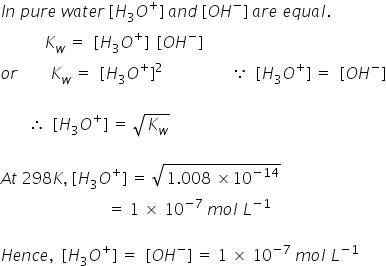Question Video: Calculating the Concentration of H3O+ Ions in an Aqueous Solution of Formic Acid | Nagwa
![SOLVED: Determine [H3O+] of a 0.230 M solution of formic acid (Ka=1.8×10−4). answer found I just need the pH**** SOLVED: Determine [H3O+] of a 0.230 M solution of formic acid (Ka=1.8×10−4). answer found I just need the pH****](https://cdn.numerade.com/ask_previews/735923cd-1fc4-40d6-8558-35b4342d62d7_large.jpg)
SOLVED: Determine [H3O+] of a 0.230 M solution of formic acid (Ka=1.8×10−4). answer found I just need the pH****

Assertion: Ammonia shows a trigonal pyramidal molecular structure.Reason: In the structure of ammonia, three atoms are attached to the central atom and thus, shows tetrahedral electron pair geometry.