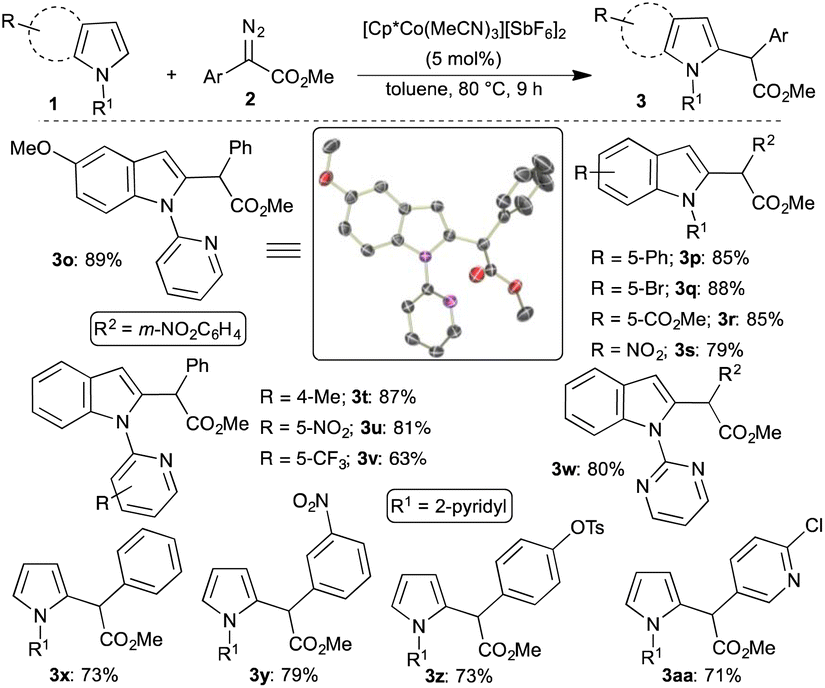
Pentamethylcyclopentadienyl)cobalt(III)‐Catalyzed C–H Bond Functionalization: From Discovery to Unique Reactivity and Selectivity - Yoshino - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library

I Have A Bowl With A Stamp Of A Crown With C.P C9 1 34 Under It. Anyone Kno... | My Antique Furniture Collection
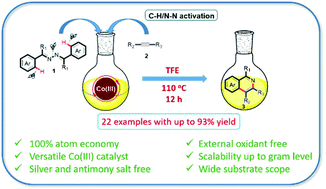
Cp*Co(iii)-catalyzed annulation of azines by C–H/N–N bond activation for the synthesis of isoquinolines - Organic & Biomolecular Chemistry (RSC Publishing)

Redefining the Mechanistic Scenario of Carbon−Sulfur Nucleophilic Coupling via High‐Valent Cp*CoIV Species - López‐Resano - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Cp*Co(III)-Catalyzed C–H Hydroarylation of Alkynes and Alkenes and Beyond: A Versatile Synthetic Tool | ACS Omega

Cp*Co(CO)I2 as Catalyst for Ortho-C,H Activation at Ferrocene: ODG-Dependent Preference for 2-Mono- or 2,5-Dialkenylation | Organometallics