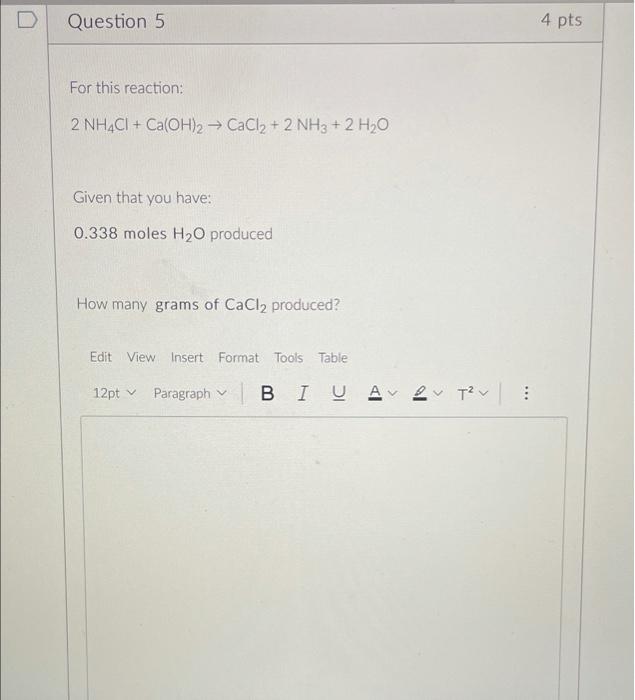
Using the formula BaO + H2SO4 = BaSO4 + H2O if 2.9 miles of water is formed during this reaction then how - Brainly.com

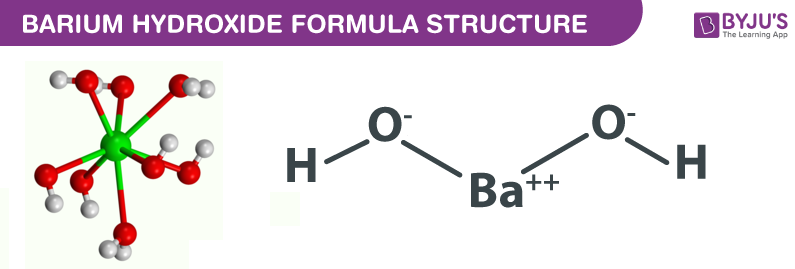
SOLVED: Organize the following in order of increasing vapor pressure: H2 02 HzO BaO H2<H2O<O2<BaO BaO<HzO < O2<Hz H2<02<HzO<BaO BaO<H2<02<HzO H2<02<BaO<HzO If the process in question 2 (a reaction has AHS rxn = -
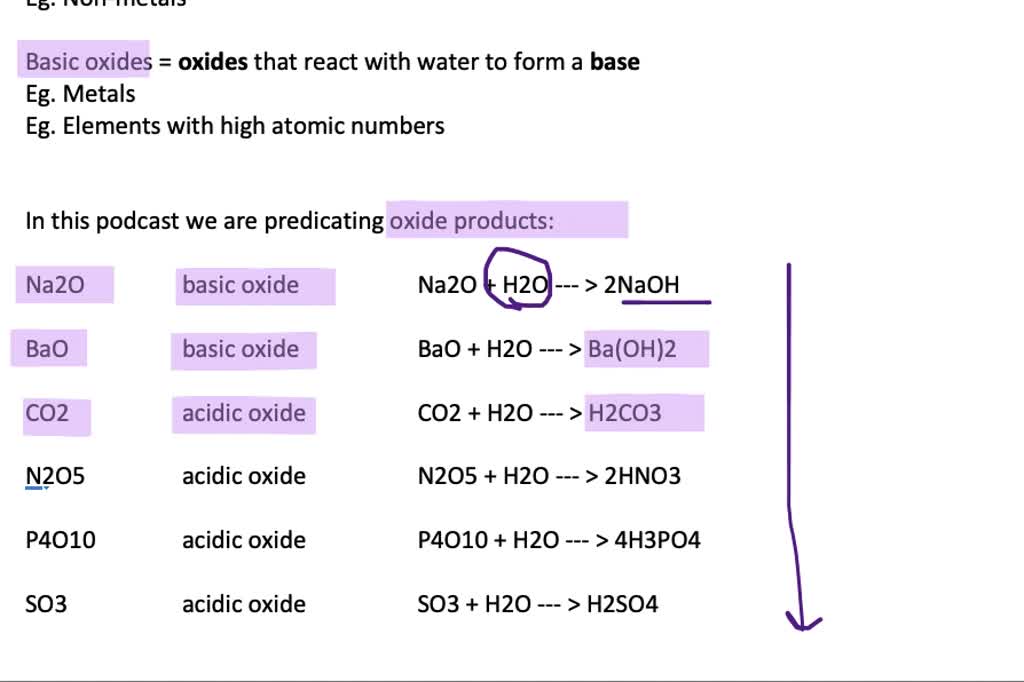
SOLVED:Predict the products of the following oxides with water: Na2 O, BaO, CO2, N2 O5, P4 O10, SO3 . Write an equation for each of the reactions. Specify whether the oxides are

How to Balance BaO + H2O = Ba(OH)2 | BaO + H2O = Ba(OH)2 balance| Chemical equation BaO + H2O - YouTube

How to Balance BaO + H2O = Ba(OH)2 | BaO + H2O = Ba(OH)2 balance| Chemical equation BaO + H2O - YouTube
![PDF] An Experimental Evaluation of Fixed and Fluidized Beds of Zeolite 13X for the Application of Compact Thermal Energy Storage | Semantic Scholar PDF] An Experimental Evaluation of Fixed and Fluidized Beds of Zeolite 13X for the Application of Compact Thermal Energy Storage | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0cf543605cbca5ca41dd21b60e732604cb37a8a9/18-Table1.1-1.png)