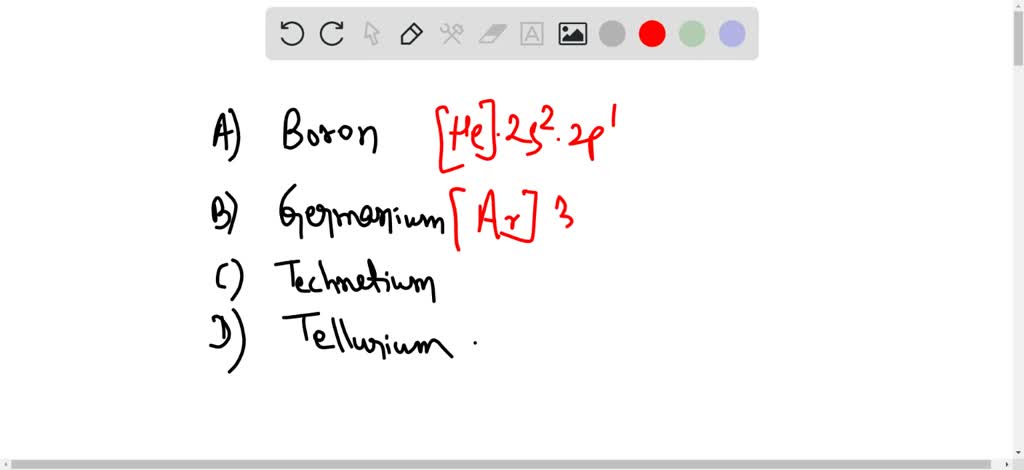
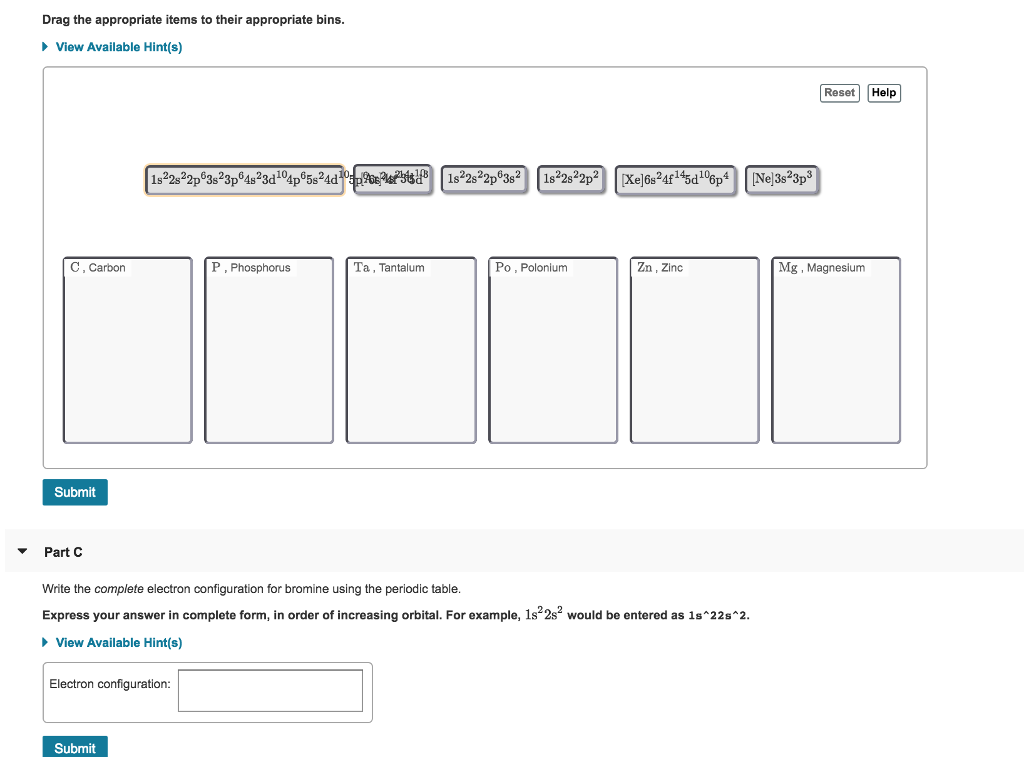
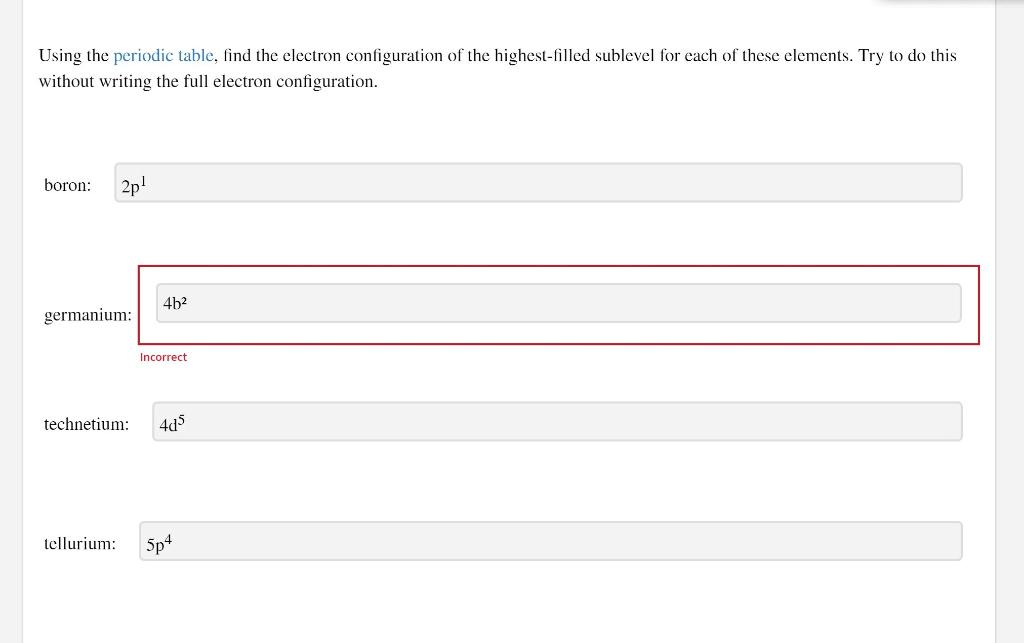
Chemistry - Notes for Final : Electric Notation : Principal Energy Level : Describethedistance of - Studocu

SOLVED:The electron affinities, in kJ / mol, for the group 1 B and group 2 B metals are as follows: (a) Why are the electron affinities of the group 2 B elements